Check out these photos of EPFL’s SwissTech Convention Center, which has a facade covered in dye-sensitized solar cells (DSSCs). The archetypal dye for a DSSC is a ruthenium complex called “black dye.” Dyes like actually have an enchanting purple-brown-black color, which isn’t totally black. But you can theoretically use any molecule that will inject an electron into a semiconductor when hit by light, and I just did a bit of hunting around and found a company called Dyenamo that specializes in different colors. The bottom picture is from C&EN, which has an article on non-silicon solar cells this week.
Author Archives: joshuagallaway
New PNNL paper on zinc/manganese oxide energy storage
I’ve had quite a few people email asking what I think about the new paper Reversible aqueous zinc/manganese oxide energy storage from conversion reactions in Nature Energy, by authors at PNNL. Understandable, since I spend a lot of my time talking about Zn and MnO2 for electrical storage. Fair!
First: unfortunately the work is being marketed (by PNNL) with a terrible graphic of a Platonic ideal supergreen™ battery that sits in a sunlit field and emits rays of light that save the world, but that’s pretty standard for battery research these days. Once the PR department gets ahold of it, you’re waist-deep in pictures of suns, windmills, iPhones, and Teslas. Most people, even most scientists, don’t understand the many levels of hierarchy involved in battery design and engineering, so I try to overlook these kinds of silly Photoshop excursions.
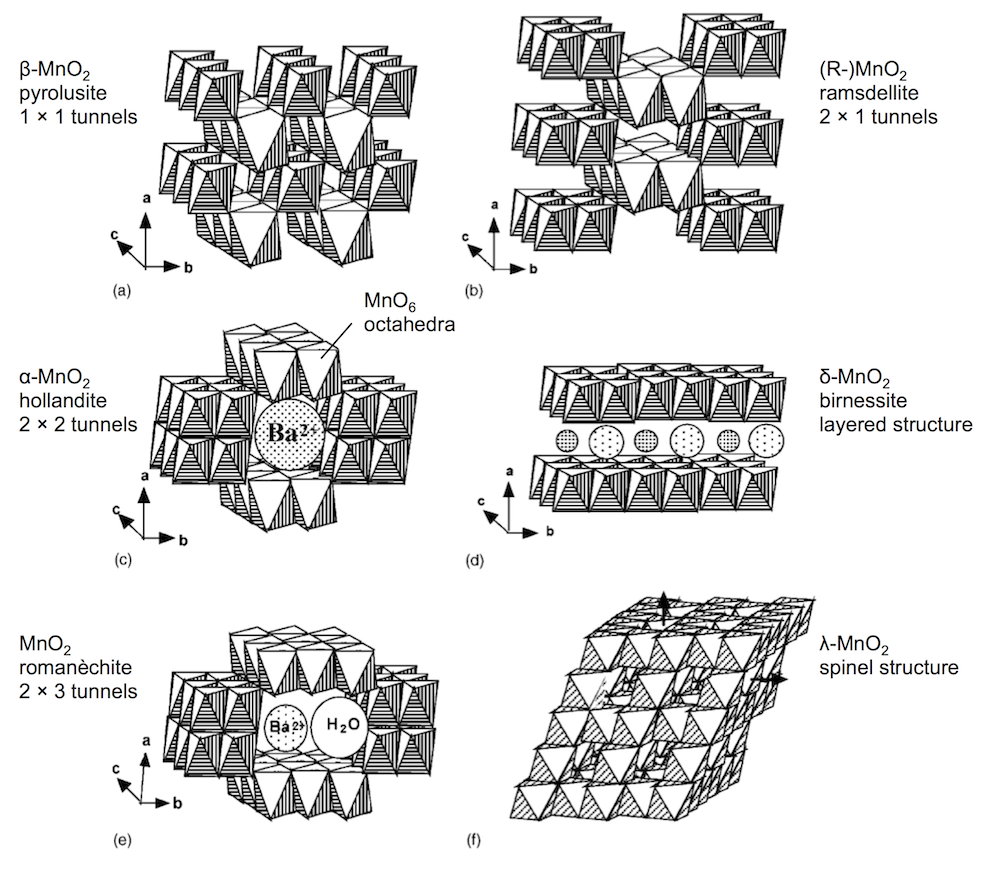
Second: the innovation of the paper is that they are making a rechargeable Zn-MnO2 battery in a mildly acidic electrolyte, and getting good cycle life. The way they’re doing this is by using α-MnO2 as their cathode active material. MnO2 comes in several polymorphic forms, some of which you can see below. (I adapted this figure from a paper by Poinsignon.) They are built from MnO6 octahedra, but can distinguished by the tunnel structures in the crystal.
A lot of my recent work has focused on the polymorph γ-MnO2, which is an intergrowth of (a) and (b) above. The PNNL work makes an interesting discovery about α-MnO2: they see the α-MnO2 going through a conversion reaction to MnOOH, which is somewhat unexpected. As you can see in the figure above, α-MnO2 is usually thought of as a host structure, to intercalate guest ions (like Ba2+). They then see that the surface of the MnOOH is coated with a large flake-like material that originates with the sulfate electrolyte, ZnSO4[Zn(OH)2]3 · xH2O. In this respect, the reaction is a bit reminiscent of a lead-acid battery, which also involves a sulfate film.
The paper is very interesting in that it provides unexpected evidence of α-MnO2 acting in the manner of a conversion reaction. (And that’s why that term is important and shows up in the paper’s title.) Also the zinc hydroxyl sulfate flake-film is a tantalizing look at what could be a very complex cathode reaction. And I’m a sucker for complex electrochemical reactions, as I hope you know. The test bed for the research was a CR2032 form factor, which is the kind of battery that goes in my running watch. So, the picture the PR machine and the science press are painting (with that world-saving battery up above) is a bit overblown, but the electrochemistry research underpinning this paper is extremely interesting, and I hope to see more.
Reactions at the single-molecule level
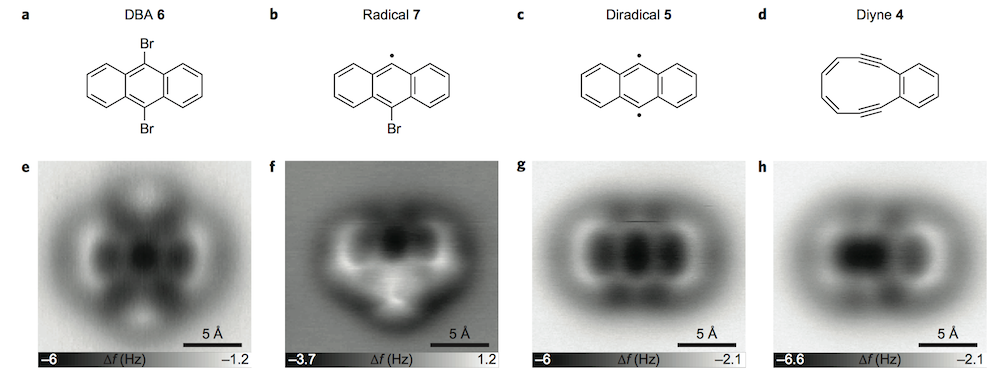
I’m always amazed by these studies where AFM and STM are used to watch cyclic molecules through a reaction. Here it’s used to image Bergman cyclization.
Battery testing at the Advanced Photon Source
Just returned from some battery testing at the Advanced Photon Source near Chicago. Here Snehal models one of the new batteries we’ve developed for grid-scale electrical storage.
And here’s a couple of them sitting in the path of an X-ray “white beam” that will give us diffraction data while they cycle. This way you can track the mechanism of what’s happening during battery cycling … since the cycling is ultimately an electrochemical transformation.
Now we’re going through a full set of data, getting to the bottom of our underlying mechanisms. By the way it snowed quite a bit while we were there. Here’s the lovely winter view from the guest house.
Manganese dioxide: the almost perfect cathode
The year is ending, and I’m wrapping up some researching findings for publication. And some exciting news: once the new year starts I’m headed to NSLS-II to use the cutting edge submicron resolution spectroscopy beamline for some experiments on a brand new battery chemistry.
First I’d like to pause to reflect on what I’ve spent the last couple years on: shallow-cycled manganese dioxide (MnO2) as a cathode material. Specifically, why we need it:
- It’s extremely cheap
- It’s extremely safe
- It’s found all over the world
- It works in aqueous (water-based) batteries
These facts make it ideal for an emerging battery market, which is large-scale, grid-level battery storage for buffering solar and wind power. This storage market will be fundamentally different than the last major new market, which emerged in the 90s: that for portable computing and electronics. Portable computing requires high energy density, with cost and safety being secondary. In contrast, the planet-wide battery deployment needed for a green (i.e. solar) future will live and die on battery cost and safety, with energy density being secondary.
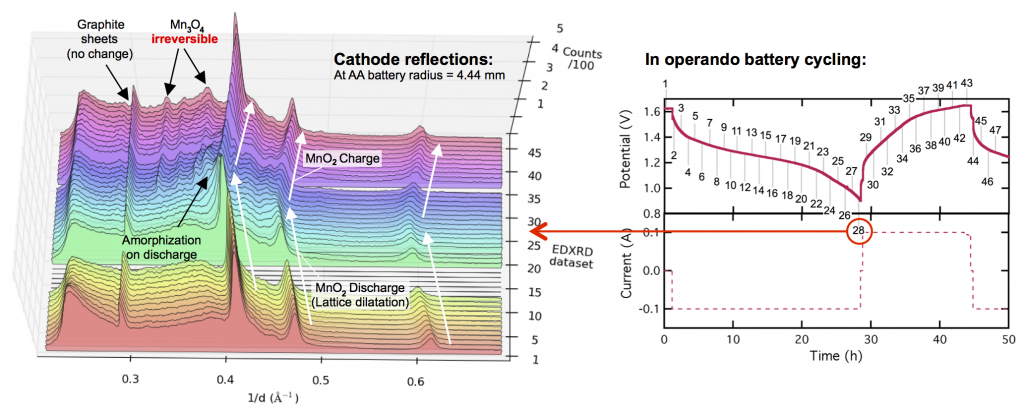
This leads to the almost in the title of this post: MnO2 is perfect for grid-scale battery storage (it even has high energy density) except its crystal structure breaks down at the end of discharge. This is shown in the X-ray diffraction data above. The three prominent MnO2 reflections shift to lower values of 1/d as the electrode discharges, because protons are inserted into the lattice causing it to expand or dilatate. Reaching the end of discharge, these reflections spread out and become dull (or somewhat amorphous) as other manganese oxides form, including the major discharge product groutite (α-MnOOH). Upon charge, groutite is converted back to MnO2, and the lattice shrinks as protons are de-inserted. We’ve gotten the MnO2 lattice back, but the problem is the “other” manganese oxides that formed at the end of discharge and are irreversible. Some reflections of hausmannite (Mn3O4) are now found in the electrode. Haumannite is highly resistive, and if too much forms, the electrode will fail. It doesn’t take many cycles. A few like this will do.
With funding from ARPA-E we developed a Zn-MnO2 battery that can cycle thousands of times, by limiting the discharge depth and not going all the way to 0.9 volts, as was done above. (That of course raises cost, but since zinc and manganese dioxide are cheap, the economics still come out in your favor.) Even then, we found that Mn3O4 (and its relative ZnMn2O4) still ends up forming around the MnO2 particles, giving them a resistive coating. Thankfully this doesn’t kill the battery, but it does mean there is a limit to how deeply you can discharge this way. However, by doing some very fundamental forensic-type experiments, resolving the manganese oxide crystal structures within undisturbed batteries, we learned something interesting. There are some important differences between the manganese oxides you see in operando (like those above) and the ones in a battery that has aged a while. In other words, the Mn3O4 seen above isn’t completely formed yet, and that suggests that perhaps its formation can be reversed, at least before too much time has passed. And that … is what we’ve been trying. Stay tuned for more on that.